The global micro-electronic medical implants market is expected to reach US$ 59,561.4 Mn in 2027 from US$ 27,461.2 Mn in 2018. The market is estimated to grow with a CAGR of 9.4% from 2019-2027.
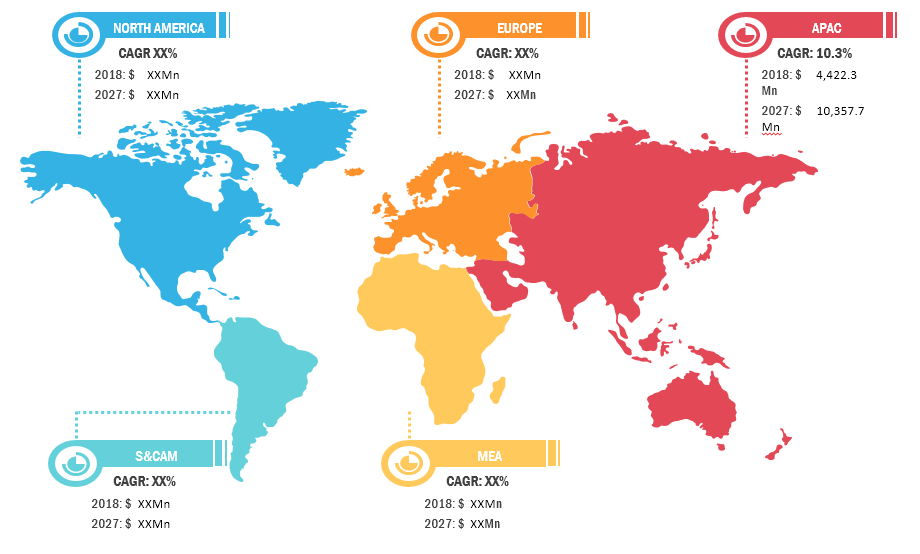
The Asia Pacific micro-electronic medical implants market is expected to grow at highest rate during the forecast period owing to higher number of research & development and increasing investments on micro-electronic medical implants development.
The research report on Micro-Electronic Medical Implants Market 2027 keenly analyzes significant features of the industry. The analysis servers market size, latest trends, drivers, threats, opportunities, as well as key market segments. It is based on past data and present market needs. Also, involve distinct business approaches accepted by the decision makers. That intensifies growth and make a remarkable stand in the industry. The Micro-Electronic Medical Implants Market will grow with a significant CAGR Between 2019 to 2027. The report segregates the complete market on the basis of key players, geographical areas, and segments.
Ask for a Sample of this Report Now at: https://www.theinsightpartners.com/sample/TIPHE100001242/
Key Benefits:
Some of the Major Players In Micro-Electronic Medical Implants Market:
An exclusive Micro-Electronic Medical Implants Market research report created through broad primary research (inputs from industry experts, companies and stakeholders) and secondary research, the report aims to present the analysis of Global Micro-Electronic Medical Implants Market By Type, By Application, By Region – North America, Europe, South America, Asia-Pacific, Middle East and Africa. The report intends to provide cutting-edge market intelligence and help decision makers take sound investment evaluation. Besides, the report also identifies and analyses the emerging trends along with major drivers, challenges and opportunities in the global Micro-Electronic Medical Implants Market. Additionally, the report also highlights market entry strategies for various companies across the globe.
The micro-electronic medical implants market by product is segmented into pacemakers & defibrillators, neurostimulators, implantable drug pumps, cochlear implants, ocular implants, retinal implants, and others. In 2018, the pacemakers and defibrillators segment held a largest market share of 32.0% of the micro-electronic medical implants market, by product. This segment is also expected to dominate the market in 2027 due to the increasing incidence of cardiovascular diseases and rise in cardiac-implant procedures. On the other hand, the cochlear implants segment is anticipated to witness the fastest growth rate of 10.1% during the forecast period, 2019 to 2027 owing to the rising demand of products and technological development in hearing aids and implant technology.
Strategic Insights
Product approvals and agreements were observed as the most adopted strategy in global micro-electronic medical implants industry. Few of the recent product key developments are listed below:
2019: BIOTRONIK announced FDA approval of the Acticor and Rivacor high-voltage cardiac rhythm management (CRM) device families for treatment of patients with cardiac arrhythmias. The six new tachycardia solutions include Rivacor VR-T, Rivacor DR-T, Rivacor HF-T QP, Acticor DX, Acticor CRT-DX Bipolar and Acticor CRT-DX.
2018: Medtronic received United States Food and Drug Administration (FDA) approval for its HVAD System. HVAD System is the smallest commercially available left ventricular assist device (LVAD) for patients with advanced heart failure. It is the only LVAD approved in the U.S. for implant via thoracotomy.
2017: Cochlear Ltd announced its strategic collaboration with Sensorion a biotech company. The collaboration will evaluate therapeutic approaches using SENS-401 in combination with cochlear implants. This collaboration has the potential to be transformational for both partners, as well as for patients suffering from hearing loss
MICRO-ELECTRONIC MEDICAL IMPLANTS – MARKET SEGMENTATION
By Product
By Technology
By Material
By Application
By End User
By Geography
Direct Purchase a copy of this report at: https://www.theinsightpartners.com/buy/TIPHE100001242/
About Us
The Insight Partners is a one stop industry research provider of actionable intelligence. We help our clients in getting solutions to their research requirements through our syndicated and consulting research services.
We are committed to providing highest quality research and consulting services to our customers. We help our clients understand key market trends, identify opportunities, and make informed decisions by providing market research solutions at an affordable cost.