
 Widespread efforts to raise awareness have succeeded, with the vast majority of women in developed nations undergoing routine breast cancer screenings, involving palpation (physical examination) and/or mammography. Unfortunately, both screening processes produce highly inaccurate results that take a personal and financial toll. Upwards of 20% of breast lumps found through palpation are not studied further, and turn out to be malignant and possibly deadly.
Widespread efforts to raise awareness have succeeded, with the vast majority of women in developed nations undergoing routine breast cancer screenings, involving palpation (physical examination) and/or mammography. Unfortunately, both screening processes produce highly inaccurate results that take a personal and financial toll. Upwards of 20% of breast lumps found through palpation are not studied further, and turn out to be malignant and possibly deadly.
The Non Invasive Diagnostic System (or NIDS) reduces the risk of misdiagnosis and increases the chance of early detection of breast cancer. Our innovative device detect 95% of all cancer, a 30% improvement over traditional mammography.
 NIDS is a non-invasive, painless, and radiation-free diagnostic device. It can be operated by an easily-trained technician in less than 15 minutes in any standard medical office setting. This ease of use creates a substantial cost-savings for both doctors and patients when compared to current diagnostic methods.
NIDS is a non-invasive, painless, and radiation-free diagnostic device. It can be operated by an easily-trained technician in less than 15 minutes in any standard medical office setting. This ease of use creates a substantial cost-savings for both doctors and patients when compared to current diagnostic methods.
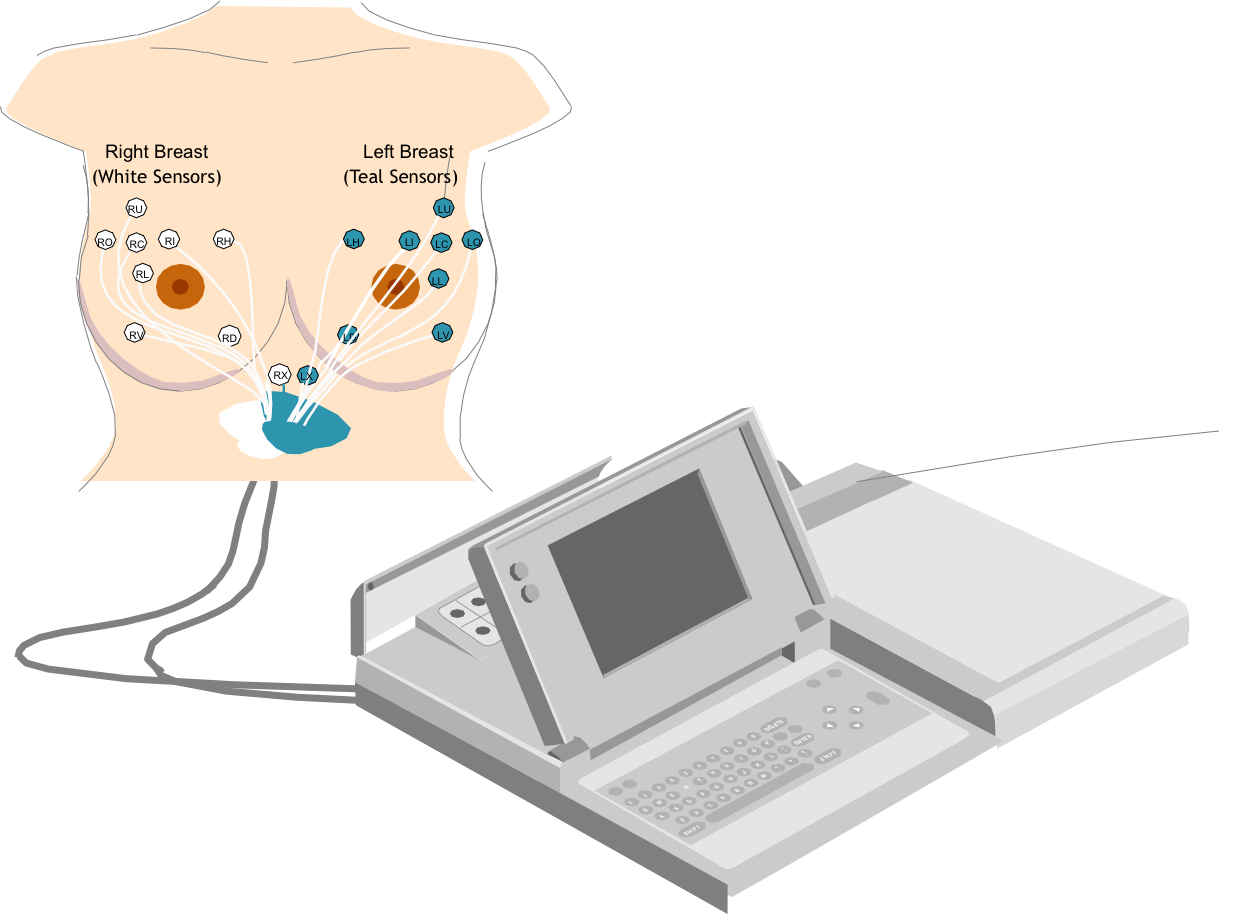
NIDS also provides physiological information that helps physicians characterize ambiguous breast findings after palpation or mammography. Prior to NIDS, these findings typically required a painful and invasive breast biopsy in order to determine malignancy.
NIDS can determine whether or not ambiguous lesions are potentially malignant without mammography or biopsy. Our technology can eliminate 55% of unnecessary biopsies in women 55 and younger.

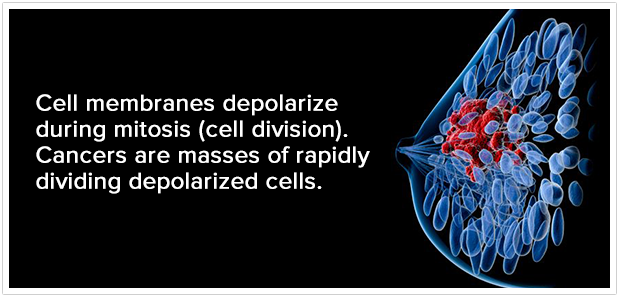
NIDS determines the biological activity of breast lesions (areas of concern that can potentially be cancerous) by utilizing a non-invasive measurement of differentials in breast tissue through sensors that are attached to a patient.
From there, a comparison is made between resting polarized cells (healthy) and rapidly dividing, depolarized cells (potentially cancerous). A NIDS Diagnostic Score compares the activity of an unaffected portion of the breast with one that may contain cancerous cells.

 Detects 95%+ of cancer (a 30% improvement over mammography)
Detects 95%+ of cancer (a 30% improvement over mammography)
 Determines whether or not ambiguous lesions are potentially malignant without mammography or biopsy
Determines whether or not ambiguous lesions are potentially malignant without mammography or biopsy
 Non invasive, radiation-free and painless
Non invasive, radiation-free and painless
 Eliminates 50% of equivocal results which lead to unnecessary biopsies (a 40% improvement over mammography)
Eliminates 50% of equivocal results which lead to unnecessary biopsies (a 40% improvement over mammography)
I n-office procedure that only takes 15 minutes and produces objective results
n-office procedure that only takes 15 minutes and produces objective results

The challenge for any diagnostic test is that it is very difficult to simultaneously achieve both high sensitivity (a measure of a test’s ability to identify the presence of disease) and high specificity (a test’s ability to identify the absence of disease). Usually a threshold is defined that represents the best clinical trade-off between sensitivity and specificity. If a test is sensitive enough (>90%), negative results can be used to safely rule out the presence of disease. Positive results associated with a high specificity test are used to rule in the presence of disease.
Neither of the major breast screening processes — palpation or mammography – is a rule-out procedure. Both require additional and often invasive testing. Typically, mammography’s sensitivity rate is 60-70% in younger women, and can exceed 80% for older women. With these sensitivity rates, mammography misses many malignant lumps, especially in younger women. At the same time, its 14% specificity rate leads to upwards of 86% unnecessary biopsies.
Seven worldwide clinical studies involving over 1,600 women have proven that NIDS was able to detect between 90% and 100% of those malignancies, a 30% improvement over mammography. These findings have been documented in several peer-reviewed journals.
We have significantly reduced the need for women being referred for additional costly tests (including biopsies) by more than half, representing a 40% improvement over mammography. The NIDS greatly decreases the occurrence of needless, painful and invasive biopsies, granting women of all ages unprecedented peace of mind.