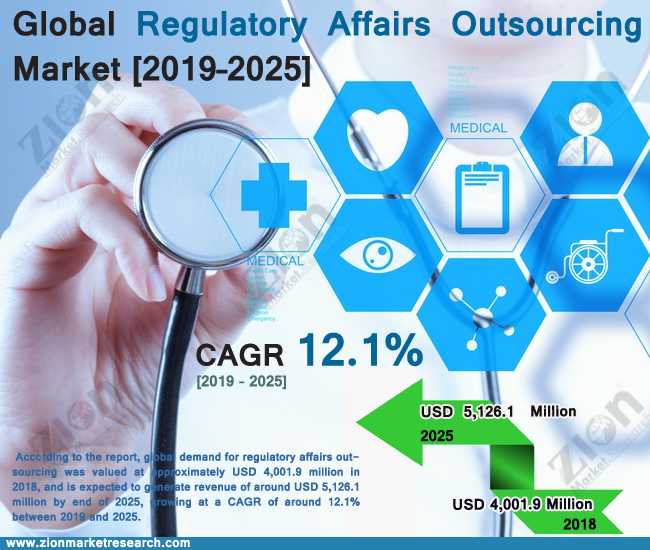
Zion Market Research has published a new report titled “Regulatory Affairs Outsourcing Market by Services (Regulatory Consulting, Legal Representation, Regulatory Writing & Publishing, Product Registration & Clinical Trial Application, and Other Regulatory Services): Global Industry Perspective, Comprehensive Analysis and Forecast, 2019 – 2025” According to the report, global demand for regulatory affairs outsourcing market was valued at approximately USD 4,001.9 million in 2018, and is expected to generate revenue of around USD 5,126.1 million by end of 2025, growing at a CAGR of around 12.1% between 2019 and 2025.
Outsourcing regulatory affairs to contract research organizations exercises provides reduced regulatory submission time, cost efficiency, and quality control in the manufacturing process. These components are expected to drive the growth over the time frame of the conjecture. Large organizations tackle capacity issues and absence of knowledge in their inner departments by outsourcing to smaller mid-sized enterprises. It also wipes out the remaining additional burden and combines administrative offices to avoid duplication of job. Regulatory affairs include exercises such as approvals drug shipment for clinical trials, drug master documents, labeling, and technical writing, reporting of severe adverse events, chemistry assessment, maintenance of Investigational New Drug (IND), eCTD conversion, query and controls management (CMC).
Request For Free Sample Report @ https://www.zionmarketresearch.com/sample/regulatory-affairs-outsourcing-industry
Increased R&D operations especially in the life science sector are expected to drive development by decreasing the general approval process by decreasing delays in legislative filings and improving Return on Investment (ROI) and cost effectiveness. In addition, favorable legislative mandates are expected during the forecast period to drive adoption. In August 2018, for example, the U.S. ICH E17 guidance on multi-regional clinical trials has been released by the Food and Drug Administration (FDA). It is anticipated that this initiative will introduce a single protocol to worldwide clinical trials, presented by various officials in distinct areas. Outsourcing of regulatory affairs has experienced few significant occurrences in the sector. In August 2018, for instance, U.K. based Syneos Health acquired Kinapse, Pharmacovigilance consulting and post-market regulatory services provider, was purchased. The firm seeks to double its consulting footprint in Europe and related business services with this purchase.
On the basis of service the market for regulatory affairs outsourcing is segmented into publishing & regulatory writing, legal representation, clinical trial application and product registration, regulatory consulting, and other regulatory services. Regulatory writing and publication retained biggest share of the market in 2018 and is likely to lead continue to lead. However, legal representation over the forecast period is expected to grow at high CAGR. Increasing demand for legal representatives in developed regions like North America and Europe are gaining traction for market permission ideas and businesses that want to set up a base in other nations concerned.
Europe, North America, Latin America, Asia Pacific, and the Middle East and Africa are the major regional segments of the global regulatory affairs outsourcing market. North America is anticipated to contribute significantly to worldwide market development. Global pharmaceutical and life science businesses, solid regulators like FDA, and talent pool accessibility will lead to overall development. In the coming years, Asia Pacific is expected to be the fastest growing region.
Some of the players included in the regulatory affairs outsourcing market are ICON Plc, Accell Clinical Research, Pharmaceutical Product Development (PPD) LLC, PARAXEL International Corporation, Covance, Inc., Genpact Ltd., Charles River Laboratories International, Inc., Medpace, Inc., Wuxi AppTec, and Criterium, Inc.
This report segments the global regulatory affairs outsourcing market as follows:
Global Regulatory Affairs Outsourcing Market: Services segment Analysis
Global Regulatory Affairs Outsourcing Market: Regional Segment Analysis
Inquire more before buying this report @ https://www.zionmarketresearch.com/inquiry/regulatory-affairs-outsourcing-industry