
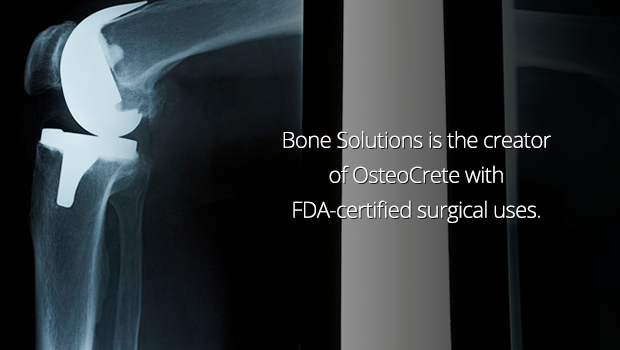
OsteoCrete is designed to improve clinical outcomes in many complex procedures like the repair of complex fractures and joint replacement procedures, and to significantly reduce the costs of these procedures.

Today’s calcium-based bone void fillers and cements often result in low surgical success rates because because they contain polymers, which frequently cause the body to reject the cements and fillers. These frequent rejections often require a second or even third follow-up surgery to correct.
OsteoCrete is a patented platform technology powder which, when added to a saline liquid, can be either injectable or moldable to fit the needs of the surgery. When it’s applied to the spaces where bone is missing, like fractures, infected areas, or areas where a tumor has been removed, OsteoCrete bonds to the existing bone to fill the void. When it’s in place, the material is absorbed by the body as the healing bone slowly replaces it.
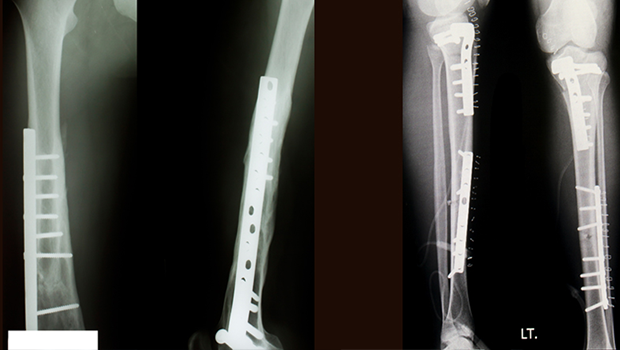
Plus, OsteoCrete hardens in less than 20 minutes, significantly shortening procedure times. Today’s calcium-based fillers can take up to 12 hours to fully harden.

 It’s a simple proposition: OsteoCrete has the most powerful profile of performance characteristics in its product category. No other bone filler can stimulate the healing of large bone defects and adhere ligaments and tendons to bone with the success of OsteoCrete.
It’s a simple proposition: OsteoCrete has the most powerful profile of performance characteristics in its product category. No other bone filler can stimulate the healing of large bone defects and adhere ligaments and tendons to bone with the success of OsteoCrete.
From Dr. Scott Rodeo published research, Hospital for Special Surgergy, New York:
“…the results of this study indicate that a magnesium-based bone adjesive can improve tendon-to-gone healing…”; “We found that this material (OsteoCrete) can induce fibrocartilage formation, limit fibrous tissue formation at the healing tendon-gbone interface, and increase osteointegration at 6 weeks,”; “It may eventually be possible to use this bone adhesive clinically to augment tendon-to-bone healing, potntially leading to increased attahment strength and a diminished risk of graft failure or slippage.”
Source: Augmentation of Tendon-to-Bne Healing With a Magnesium-Based Bone Adhesive, by L. Gulotta, MD, D Kovacevic, L. Ying, J. Ehteshani, MD, S. Montgomery, and Scott A;. Rodeo, MD From the laboratory for Soft Tissue Research, Hospital for Special Surgery, New York, The American Journal of Sports Medicine, Vol. 36, No. 7, 2008.
The results Bone Solutions believes will be a drastically increased success rate in procedures that are notoriously difficult to perform: joint replacements and orthopedic surgeries needed as a result of trauma.
From published research by Joseph P. Iannotti, MD, Ph.D., Head of Orthopedics Department, The Cleveland Clinic, Cleveland, OH:
“We demonstrated in this study that OsteoCrete was very comparable to PMMA bone cement when tested in tension for initial stiffness, failure load, failure displacement, failure cycle and total work.”; “OsteoCrete is osteoconductive, biocompatible and resorbs over time while supporting bone formation, whereas PMMA does not have these properties.”; This study demonstrates that OsteoCrete has short term fixation similar to that of PMMA bone cement and may therefore provide sufficient mechanical support of a metaphysical implant in patients with poor bone quality where augmentation of implant fixation is desired.”
Source: Can a Bone Graft Substitute Provide Initial Fixation for Humeral Implant in Cancellous Bone?, by Joseph P. Iannotti, MD, Ph.D.,, Myung-Sun-Kim, MD, Ph.D., David Kovacevic, MD., Bong-Jae Jun, Ph.D., Katherin A Derwin, Ph.D., Cleveland Clinic Foundation, Cleveland, OH, United States

For clients, for doctors, for insurance companies
 OsteoCrete’s unique capabilities give it the potential to be the one material that achieves rising market share from existing bone void filler and bone cement products.
OsteoCrete’s unique capabilities give it the potential to be the one material that achieves rising market share from existing bone void filler and bone cement products.
 Because of the significantly higher success rates, OsteoCrete reduces the requirement for costly follow-up surgeries.
Because of the significantly higher success rates, OsteoCrete reduces the requirement for costly follow-up surgeries.
 The industry currently spends hundreds of millions of dollars in R&D to invent better calcium-based products, or to invent devices like orthopedic pins, plates, and screws that better reinforce existing bone structure (these materials, in addition, are not bioabsorbable).
The industry currently spends hundreds of millions of dollars in R&D to invent better calcium-based products, or to invent devices like orthopedic pins, plates, and screws that better reinforce existing bone structure (these materials, in addition, are not bioabsorbable).

 Patients
Patients
 less time under anesthesia
less time under anesthesia
 fewer days in hospital
fewer days in hospital
 100% replaced by patient’s bone
100% replaced by patient’s bone
 more natural resorption rates
more natural resorption rates
 less pain, less need for pain medication
less pain, less need for pain medication
 Surgeons
Surgeons
 easier to implant
easier to implant
 better fracture reduction
better fracture reduction
 100% replaced by patient’s bone
100% replaced by patient’s bone
 less time in procedure
less time in procedure
 better visualization of X-ray
better visualization of X-ray
 improved outcomes
improved outcomes
 Healthcare (hospitals, health insurance companies, etc.)
Healthcare (hospitals, health insurance companies, etc.)
 lower operating room times
lower operating room times
 reduced hospital stay
reduced hospital stay
 higher operating room productivity
higher operating room productivity
 less need for multiple treatments
less need for multiple treatments
 earlier, faster recovery
earlier, faster recovery
 less need for home healthcare
less need for home healthcare

OsteoCrete has already received FDA 510(K) clearance as the world’s first and only magnesium-based bone void filler. The technology behind OsteoCrete is protected by a seminal U.S. patent, and we’ve filed an additional four patents to protect our innovation.
Recently, we were proud to be the recipient of the North American Bone Graft Technology Leadership of the Year Award.
OsteoCrete is a final product, and has been fully tested for both safety and efficacy. Our official launch will come in the fall of 2015, and by then, we expect to disclose the details of our first partnership with a global distribution company ranked in the top 10 in size in the global orthopedics industry.
Strategic Worldscale Third Party Relationships are yet another value added milestones that Bone Solutions has achieved for its potential investors. In hand is a term sheet for global distribution for the global extremity market with one of the largest firms in the world in this segment. Next, a Material Transfer Agreement (MTA) has been signed with a top five global leader in the spine market. In addition are term sheet discussions with a Cranial Maxillofacial implant company, and then a Material Transfer Agreement with a major US dental implant company, along with an NDA now signed with a premiue foreign dental implant company.

 Tom is one of three Co-Founders of Bone Solutions and is also CEO of Bindan Corporation, his parent technology company focused on road/bridge repair solutions using his binder technology, that also holds equity in BSI. Tom is designer and inventor of OsteoCrete. Tom other affiliates are two other technology companies: a manufacturer of ADA detectable warning tiles and a manufacturer of oil well cements. He holds eight patents from the USPTO. Tom’s binder technology for road repair products was a foundation for this Bone Solutions OsteoCrete medical applications.
Tom is one of three Co-Founders of Bone Solutions and is also CEO of Bindan Corporation, his parent technology company focused on road/bridge repair solutions using his binder technology, that also holds equity in BSI. Tom is designer and inventor of OsteoCrete. Tom other affiliates are two other technology companies: a manufacturer of ADA detectable warning tiles and a manufacturer of oil well cements. He holds eight patents from the USPTO. Tom’s binder technology for road repair products was a foundation for this Bone Solutions OsteoCrete medical applications.
 Drew as CEO of BSI, brings 26 years of experience in the healthcare field, with the last five as CEO and cofounder of Heart Test Laboratories, Inc. He was also Senior VP of Global Sales for Criticare Systems.
Drew as CEO of BSI, brings 26 years of experience in the healthcare field, with the last five as CEO and cofounder of Heart Test Laboratories, Inc. He was also Senior VP of Global Sales for Criticare Systems.
 Tony is also a Co-Founder, and was the Former VP and Manager of the Petroleum Group at Salomon Brothers in New York, and the former VP of Corporate Finance for Ray Hunt’s enterprises, namely Dallas based Hunt Oil Company, the world’s largest independent oil company for 17 years. He was also the Senior Economist for the American Petroleum Institute in Washington, D.C. Most recently he has been Managing Executive, Copp Ventures.
Tony is also a Co-Founder, and was the Former VP and Manager of the Petroleum Group at Salomon Brothers in New York, and the former VP of Corporate Finance for Ray Hunt’s enterprises, namely Dallas based Hunt Oil Company, the world’s largest independent oil company for 17 years. He was also the Senior Economist for the American Petroleum Institute in Washington, D.C. Most recently he has been Managing Executive, Copp Ventures.
 John has over 25 years of experience in marketing and business planning. He spent 30 years at IBM working as Director of Finance for several projects. He is the former Director of Marketing for Telular Corporation and Adjunct Professor of Marketing at the College of St. Francis.
John has over 25 years of experience in marketing and business planning. He spent 30 years at IBM working as Director of Finance for several projects. He is the former Director of Marketing for Telular Corporation and Adjunct Professor of Marketing at the College of St. Francis.